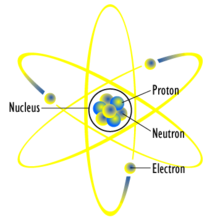
Structure of atom
Dalton was the first to devise the atomic theory to explain certain experimental facts. However the basis of his atomic theory is largely disputed.
In 1911, Ernest Rutherford gave a new atomic model which included a nuclear and orbiting electrons. His atomic theory was based on the famous experiment carried out earlier by his assistants where a thin gold foil was bombarded with alpha particles. By examining the deflection experienced by the alpha particles, he deduced that an atom has a heavy nucleus and is made up of mostly empty space.
|
Rutherford's atomic model suffers several problems theoretically. Neils Bohr proposed a theoretically new model with the following postulates:
|